STREAMLINED PHARMACEUTICAL DISPENSING AND STOCK MANAGEMENT
Achieved through automated prescription workflows, real-time inventory tracking, and barcode-integrated dispensing systems that reduce delays and errors.
LOWER OPERATIONAL COSTS
Enabled by centralized pharmaceutical dashboards, billing integration, and intelligent inventory management tools that minimize waste and overstock.
EXPERTS ON BOARD
100% of our clients scale their pharmaceutical operations with us, trusting our domain expertise, regulatory knowledge, and focus on medication safety.
PROJECTS DELIVERED
Custom pharmaceutical solutions built for manufacturers, distributors, and healthcare networks across global markets.
Custom AI Data Processing & Search System
Accelerate your lab workflows with a tailored automation solution that scans, structures, and filters research data using metadata-driven algorithms. Unlike generic off-the-shelf tools, we build systems designed around your specific data types, collection methods, and lab formats — ensuring seamless search, sorting, and cross-referencing.
From experiment logs and sensor outputs to biomedical records, our platform automatically processes incoming data and links it with internal or open-access knowledge bases, giving your team instant, contextual access to the full research picture.
Custom AI Search Engine with Natural Language Understanding (NLU)
We build an intelligent search engine that enables teams to quickly find relevant research, files, experiments, or graphs — even if they are named differently or described in another way.Such solutions integrates with internal repositories, databases, and file systems of the company and conducts searches within scientific articles and publicly available research. It analyzes content, context, and associated metadata to provide a comprehensive picture of previous studies. The system also learns over time, improving search accuracy and understanding of corporate terminology.
Custom Models for Molecule Analysis:
Developing custom AI models to analyze large datasets of chemical and biological data. These models allow researchers to predict properties of molecules such as toxicity, bioavailability, and effectiveness based on historical data and previous experiments. These solutions help significantly reduce the number of tests by focusing attention on the most promising candidates.
Custom AI-Driven In Vitro Research
Strengthen your in vitro testing processes with custom AI models that analyze and predict outcomes for a range of tests, including drug interactions with cell cultures, toxicity analysis of chemical compounds, enzymatic activity studies, and genetic or biochemical reactions in controlled environments. By leveraging both historical and real-time data, these models reduce the need for repetitive testing, streamline decision-making, and accelerate the path to reliable results.
AI for In Vivo Planning and Analysis
AI enables intelligent in vivo planning by analyzing historical and real-time data from previous experiments, optimizing testing conditions, and conducting data searches to identify the most relevant organisms and conditions for new molecule testing. This solution allows researchers to predict the organism’s reaction to new molecules without the need for physical tests. AI helps select the most promising candidates for experimentation, minimizing required tests and enhancing research efficiency.
Drug Interaction Custom Prediction Models
Utilizing AI to create predictive models that analyze chemical properties, biological effects, and historical data to predict potential interactions between different drugs. We develop custom solutions that use machine learning algorithms trained on large datasets to forecast how drugs might interact in the human body, helping healthcare providers avoid dangerous combinations.
We create personalized drug interaction prediction systems that integrate chemical, pharmacological, and patient data. By analyzing known interactions and the individual conditions of the patient, we provide healthcare providers with valuable insights that help prevent harmful effects from drug combinations.
Custom AI-Powered Data Insight Platform
A custom AI-powered data analysis platform that uses artificial intelligence to identify correlations, patterns, and hidden dependencies in your historical experimental data. This enables better understanding of processes, optimization of bioprocess experimental options, and more informed decision-making without the need for costly physical testing.
Implement an AI-powered solution that integrates clinical trial data from diverse sources, such as patient demographics, biomarkers, medical histories, treatment protocols, and clinical outcomes. The system uses machine learning algorithms to analyze historical and ongoing data, automatically recognizing patterns within different patient groups and predicting treatment outcomes. It combines data from multiple clinics and sources into a unified platform, enabling a comprehensive view of the trial, improving data integration and correlation. This allows for faster, more accurate analysis, enhances decision-making by providing visual insights, and ultimately leads to more informed choices regarding drug efficacy and patient safety.
Custom AI-Driven Regulatory Documentation Management
We propose a custom AI-assisted Regulatory Intelligence and Document Management System designed to support your team in managing compliance throughout the entire research lifecycle. The system helps monitor global regulatory sources and quickly identify data relevant to new or updated requirements. Once updates are detected, it assists in locating associated internal documents and processes that may need to be revised. The system streamlines document navigation, highlights what requires review or re-approval, and recommends next steps based on predefined workflows.
Custom Integrated Data Management Platform
Break down departmental barriers by implementing a unified, automated data management platform that ensures seamless synchronization across all R&D departments and is fully tailored to your organizational structure and needs.
We design custom solutions that consolidate data from research, experimental, laboratory, preclinical, clinical, and analytical areas into one environment. This enables faster collaboration, more accurate analytics, and full regulatory compliance.
Custom IoT Systems
Utilize an IoT-based solution to collect and analyze real-time data from lab equipment and biosensors. By integrating sensors that monitor critical variables such as pH, temperature, oxygen levels, and nutrient concentrations, this system allows researchers to track and adjust experimental conditions continuously. It provides actionable insights for optimized decision-making, automating responses to changing conditions and ensuring more efficient experiment management.
AI Predictive Maintenance
We develop a custom-tailored predictive maintenance solution specifically for pharmaceutical manufacturing, where IIoT sensors are installed on critical equipment like compressors, cleanroom systems, and sterilization units to monitor parameters such as temperature, vibration, and pressure in real time. This data feeds into an analytics engine trained on typical equipment behavior in regulated production environments, enabling early detection of potential issues and forecasting failures 7–14 days in advance. Maintenance can then be scheduled proactively during planned downtime. The solution integrates with existing production and quality systems, automatically triggering service tasks and ensuring traceable, audit-ready maintenance workflows.
Custom Vision AI System
We develop a custom Vision AI system that seamlessly integrates with existing pharmaceutical production lines to enable real-time quality control. The system automatically detects shape defects, color inconsistencies, and foreign particles in tablets, capsules, and vials; verifies label accuracy, serial numbers, barcodes, and expiration dates without stopping the line; inspects liquid fill levels and cap integrity by comparing each unit to a reference image; and removes defective units from the line using robotic arms. All data is captured and securely stored to ensure full traceability and compliance.
AI-Powered Inline Counting System
We implement vision-enabled inline counting systems that use advanced Vision AI to count pharmaceutical units — vials, ampoules, capsules, or packaging — in real time with <1% error margin. This eliminates the need for bulky traditional counters and reduces human involvement on the line. The counting data is instantly transmitted to the warehouse management system (WMS), allowing automated stock updates, triggering reorder workflows, and ensuring production continuity by preventing stockouts of critical components.
Custom Digital Compliance Platform
We create platforms that integrate with existing QMS, MES, LIMS, and ERP systems to automate regulatory change monitoring, document version control, and real-time audit-ready reporting. The platform continuously tracks global regulatory sources, flags relevant changes, and links them to affected SOPs, batch templates, and validation protocols, guiding experts through necessary updates. The e-Batch Records module automatically collects data from production lines, eliminating manual entry errors, and captures immutable audit trails and compliant electronic signatures.
Custom Smart Energy Management Platform
We develop smart energy management platforms that integrate with existing BMS, MES, ERP, and IoT infrastructures to enable real-time monitoring, automated control, and predictive energy optimization. The platform continuously collects data from HVAC systems, chillers, and process equipment via wireless sensors, analyzing it with AI algorithms to detect inefficiencies, predict performance degradation, and recommend optimal operating parameters based on production schedules and environmental conditions. Interactive dashboards provide instant visibility into energy consumption per batch or shift, while automated alerts notify teams about abnormal patterns or required maintenance, helping pharmaceutical plants reduce energy costs and improve sustainability.
Custom Digital Waste Management Platform
We build purpose-driven waste management modules that integrate with MES, LIMS, and ERP systems to automate the classification, tracking, and reporting of pharmaceutical waste. Real-time production and lab data feed into the platform to categorize waste accurately, auto-generate compliant documentation, and provide centralized dashboards for audit readiness. Integrated sustainability analytics track packaging use and support the shift toward recyclable and biodegradable alternatives.
Custom IoT Platform for Drug Delivery Control
The development of an IoT platform that integrates with existing systems to track the storage conditions and transportation of drugs at every stage of the supply chain in real-time. Sensors are used to monitor temperature, humidity, and location, allowing for prompt responses to changes and ensuring compliance with storage conditions.
Custom Pharmacovigilance System
An AI-powered platform for drug safety monitoring that uses data analytics collected from hospitals and consumer reports to identify adverse effects and enable quick responses.
Custom Patient Support System
A mobile app-based solution utilizing AI to support patients in adhering to their treatment plans. Such platforms offer companies an effective tool for promoting both existing and new products to their patient base. It provides personalized recommendations and reminders tailored to specific conditions, helping patients track their medication intake, adjust dosages, and monitor their treatment progress. Patients can upload their test results and progress, allowing for more informed treatment adjustments.
Custom AI Data Processing & Search System
Accelerate your lab workflows with a tailored automation solution that scans, structures, and filters research data using metadata-driven algorithms. Unlike generic off-the-shelf tools, we build systems designed around your specific data types, collection methods, and lab formats — ensuring seamless search, sorting, and cross-referencing.
From experiment logs and sensor outputs to biomedical records, our platform automatically processes incoming data and links it with internal or open-access knowledge bases, giving your team instant, contextual access to the full research picture.

Custom AI Search Engine with Natural Language Understanding (NLU)
We build an intelligent search engine that enables teams to quickly find relevant research, files, experiments, or graphs — even if they are named differently or described in another way.Such solutions integrates with internal repositories, databases, and file systems of the company and conducts searches within scientific articles and publicly available research. It analyzes content, context, and associated metadata to provide a comprehensive picture of previous studies. The system also learns over time, improving search accuracy and understanding of corporate terminology.

Custom Models for Molecule Analysis:
Developing custom AI models to analyze large datasets of chemical and biological data. These models allow researchers to predict properties of molecules such as toxicity, bioavailability, and effectiveness based on historical data and previous experiments. These solutions help significantly reduce the number of tests by focusing attention on the most promising candidates.

Custom AI-Driven In Vitro Research
Strengthen your in vitro testing processes with custom AI models that analyze and predict outcomes for a range of tests, including drug interactions with cell cultures, toxicity analysis of chemical compounds, enzymatic activity studies, and genetic or biochemical reactions in controlled environments. By leveraging both historical and real-time data, these models reduce the need for repetitive testing, streamline decision-making, and accelerate the path to reliable results.

AI for In Vivo Planning and Analysis
AI enables intelligent in vivo planning by analyzing historical and real-time data from previous experiments, optimizing testing conditions, and conducting data searches to identify the most relevant organisms and conditions for new molecule testing. This solution allows researchers to predict the organism’s reaction to new molecules without the need for physical tests. AI helps select the most promising candidates for experimentation, minimizing required tests and enhancing research efficiency.

Drug Interaction Custom Prediction Models
Utilizing AI to create predictive models that analyze chemical properties, biological effects, and historical data to predict potential interactions between different drugs. We develop custom solutions that use machine learning algorithms trained on large datasets to forecast how drugs might interact in the human body, helping healthcare providers avoid dangerous combinations.
We create personalized drug interaction prediction systems that integrate chemical, pharmacological, and patient data. By analyzing known interactions and the individual conditions of the patient, we provide healthcare providers with valuable insights that help prevent harmful effects from drug combinations.

Custom AI-Powered Data Insight Platform
A custom AI-powered data analysis platform that uses artificial intelligence to identify correlations, patterns, and hidden dependencies in your historical experimental data. This enables better understanding of processes, optimization of bioprocess experimental options, and more informed decision-making without the need for costly physical testing.

Implement an AI-powered solution that integrates clinical trial data from diverse sources, such as patient demographics, biomarkers, medical histories, treatment protocols, and clinical outcomes. The system uses machine learning algorithms to analyze historical and ongoing data, automatically recognizing patterns within different patient groups and predicting treatment outcomes. It combines data from multiple clinics and sources into a unified platform, enabling a comprehensive view of the trial, improving data integration and correlation. This allows for faster, more accurate analysis, enhances decision-making by providing visual insights, and ultimately leads to more informed choices regarding drug efficacy and patient safety.

Custom AI-Driven Regulatory Documentation Management
We propose a custom AI-assisted Regulatory Intelligence and Document Management System designed to support your team in managing compliance throughout the entire research lifecycle. The system helps monitor global regulatory sources and quickly identify data relevant to new or updated requirements. Once updates are detected, it assists in locating associated internal documents and processes that may need to be revised. The system streamlines document navigation, highlights what requires review or re-approval, and recommends next steps based on predefined workflows.

Custom Integrated Data Management Platform
Break down departmental barriers by implementing a unified, automated data management platform that ensures seamless synchronization across all R&D departments and is fully tailored to your organizational structure and needs.
We design custom solutions that consolidate data from research, experimental, laboratory, preclinical, clinical, and analytical areas into one environment. This enables faster collaboration, more accurate analytics, and full regulatory compliance.

Custom IoT Systems
Utilize an IoT-based solution to collect and analyze real-time data from lab equipment and biosensors. By integrating sensors that monitor critical variables such as pH, temperature, oxygen levels, and nutrient concentrations, this system allows researchers to track and adjust experimental conditions continuously. It provides actionable insights for optimized decision-making, automating responses to changing conditions and ensuring more efficient experiment management.

AI Predictive Maintenance
We develop a custom-tailored predictive maintenance solution specifically for pharmaceutical manufacturing, where IIoT sensors are installed on critical equipment like compressors, cleanroom systems, and sterilization units to monitor parameters such as temperature, vibration, and pressure in real time. This data feeds into an analytics engine trained on typical equipment behavior in regulated production environments, enabling early detection of potential issues and forecasting failures 7–14 days in advance. Maintenance can then be scheduled proactively during planned downtime. The solution integrates with existing production and quality systems, automatically triggering service tasks and ensuring traceable, audit-ready maintenance workflows.
Custom Vision AI System
We develop a custom Vision AI system that seamlessly integrates with existing pharmaceutical production lines to enable real-time quality control. The system automatically detects shape defects, color inconsistencies, and foreign particles in tablets, capsules, and vials; verifies label accuracy, serial numbers, barcodes, and expiration dates without stopping the line; inspects liquid fill levels and cap integrity by comparing each unit to a reference image; and removes defective units from the line using robotic arms. All data is captured and securely stored to ensure full traceability and compliance.

AI-Powered Inline Counting System
We implement vision-enabled inline counting systems that use advanced Vision AI to count pharmaceutical units — vials, ampoules, capsules, or packaging — in real time with <1% error margin. This eliminates the need for bulky traditional counters and reduces human involvement on the line. The counting data is instantly transmitted to the warehouse management system (WMS), allowing automated stock updates, triggering reorder workflows, and ensuring production continuity by preventing stockouts of critical components.
Custom Digital Compliance Platform
We create platforms that integrate with existing QMS, MES, LIMS, and ERP systems to automate regulatory change monitoring, document version control, and real-time audit-ready reporting. The platform continuously tracks global regulatory sources, flags relevant changes, and links them to affected SOPs, batch templates, and validation protocols, guiding experts through necessary updates. The e-Batch Records module automatically collects data from production lines, eliminating manual entry errors, and captures immutable audit trails and compliant electronic signatures.

Custom Smart Energy Management Platform
We develop smart energy management platforms that integrate with existing BMS, MES, ERP, and IoT infrastructures to enable real-time monitoring, automated control, and predictive energy optimization. The platform continuously collects data from HVAC systems, chillers, and process equipment via wireless sensors, analyzing it with AI algorithms to detect inefficiencies, predict performance degradation, and recommend optimal operating parameters based on production schedules and environmental conditions. Interactive dashboards provide instant visibility into energy consumption per batch or shift, while automated alerts notify teams about abnormal patterns or required maintenance, helping pharmaceutical plants reduce energy costs and improve sustainability.

Custom Digital Waste Management Platform
We build purpose-driven waste management modules that integrate with MES, LIMS, and ERP systems to automate the classification, tracking, and reporting of pharmaceutical waste. Real-time production and lab data feed into the platform to categorize waste accurately, auto-generate compliant documentation, and provide centralized dashboards for audit readiness. Integrated sustainability analytics track packaging use and support the shift toward recyclable and biodegradable alternatives.

Custom IoT Platform for Drug Delivery Control
The development of an IoT platform that integrates with existing systems to track the storage conditions and transportation of drugs at every stage of the supply chain in real-time. Sensors are used to monitor temperature, humidity, and location, allowing for prompt responses to changes and ensuring compliance with storage conditions.

Custom Pharmacovigilance System
An AI-powered platform for drug safety monitoring that uses data analytics collected from hospitals and consumer reports to identify adverse effects and enable quick responses.

Custom Patient Support System
A mobile app-based solution utilizing AI to support patients in adhering to their treatment plans. Such platforms offer companies an effective tool for promoting both existing and new products to their patient base. It provides personalized recommendations and reminders tailored to specific conditions, helping patients track their medication intake, adjust dosages, and monitor their treatment progress. Patients can upload their test results and progress, allowing for more informed treatment adjustments.

VP of Customer Success in Healthcare

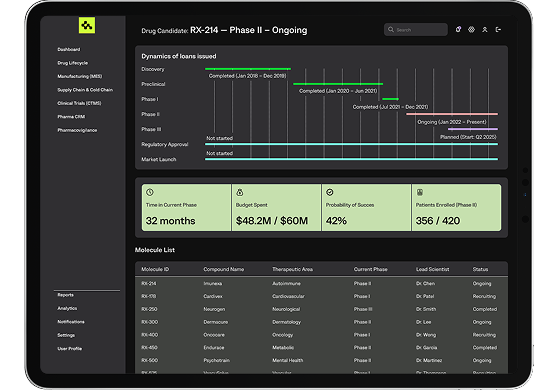
We build end-to-end platforms to manage every phase of the drug lifecycle, from R&D and clinical trials to regulatory approval and post-market surveillance, ensuring traceability, compliance, and speed to market.
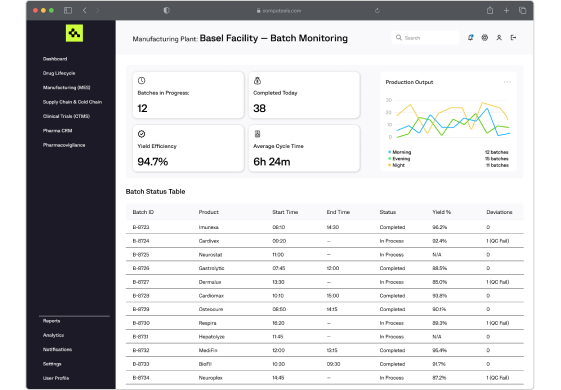
Computools develops MES solutions tailored for pharmaceutical production, enabling batch tracking, quality control, GMP compliance, and real-time monitoring of equipment and workflows.
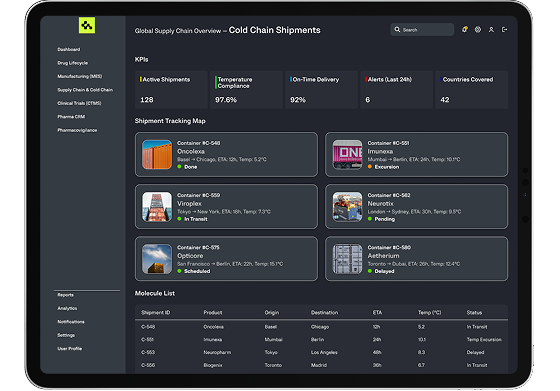
Our custom platforms optimize pharmaceutical logistics with real-time tracking, temperature monitoring, and smart routing to maintain product integrity and ensure regulatory compliance during transport and storage.
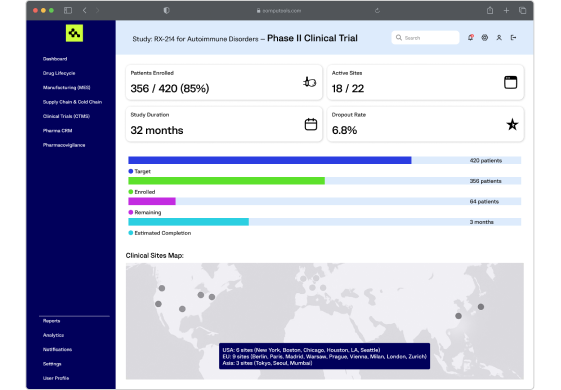
We develop custom CTMS platforms for protocol tracking, patient enrollment, site management, and regulatory submission, improving trial transparency and shortening time-to-market for new drugs.
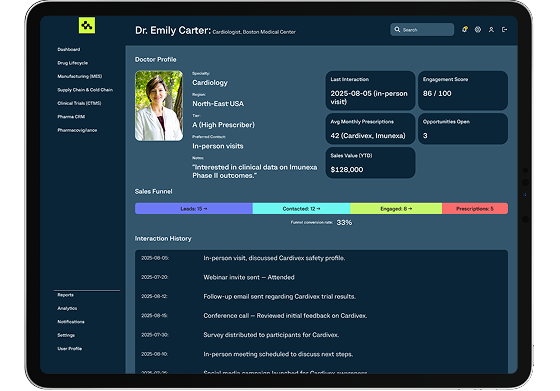
We create CRM tools for pharma reps and distributors with territory management, HCP profiling, campaign automation, and integrated reporting, driving sales and improving HCP engagement.
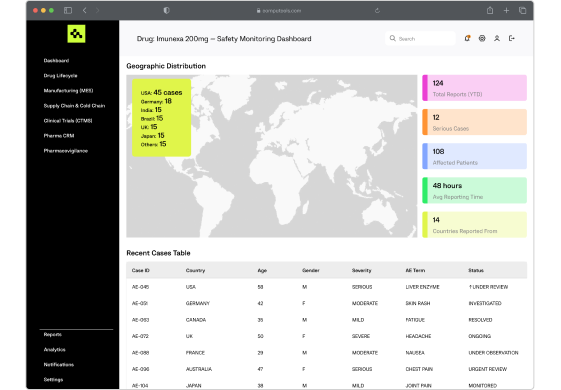
Our platforms support real-time adverse event collection, signal detection, and automated reporting to regulatory bodies, helping pharmaceutical companies maintain drug safety and compliance worldwide.
“The team was very friendly and had the highest level of competence, engagement, and project management.”

“Computools predicted all possible points of our business growth and implemented them into the project.”

“We were highly satisfied with their deep understanding of our fintech processes and their project management was really superb.”

“Computools is a highly professional company with a skilled and responsive team. Their ability to propose valuable improvements and their dedication to the project made a significant difference.”

“The most noteworthy value that stood out was their exceptional experience in developing AI software solutions.”

“We were deeply impressed with their technical expertise, transparency, and flexibility. The team was highly skilled, easy to work with, and always proactive in solving challenges.“

“Computools offered non-standard solutions and maximized their investment in our business success.“

“Our company is impressed by their client-first approach and deep niche expertise.”

“A very comfortable collaboration and clear communication on every stage of platform development and maintenance.”

“After all these years, Computools never fails to arrive on time and with a quality that never ceases to amaze me. They work well as a team and are adaptable and communicative.”

“Within the first three months of its use, the designed program by Computools significantly reduced meter reading fraud by over 30%. Additionally, we saw a rise in operational effectiveness. Customer comments highlighted greater billing transparency and speedier service delivery, which contributed to an improvement in customer satisfaction levels.”

“They were professional, adapted to our short-notice needs, documented everything, and were transparent.”

“Thanks to Computools, we have seen a 15% growth in sales and a 40% boost in user satisfaction. Our image management has become more efficient, and our diagnostic capabilities have improved. Overall, the team has delivered a high-quality solution that meets our requirements.”

“Computools has significantly improved our LMS. The team holds regular meetings and provides detailed project reports, keeping us well-informed. We communicate via email, and overall, everything has gone smoothly.”

“Computools worked closely with us to understand our challenges. They developed a platform that integrated seamlessly with our existing infrastructure and Automatic Identification Systems (AIS) to capture private vessel data.”

“Computools’ work has had a positive impact on the client’s business. The team is flexible and responsive to the client’s needs. Their expertise has been key to the project’s success. Overall, the engagement has been positive.”

“Due to the platform’s use, the new products’ generated go-to-market timeline improves by 20%, cutting down on plan costs and, most importantly, enhancing the connection between the departments. The availability of near real-time information and the enhancement of the speed of decision-making are truly remarkable.”

“Thanks to Computools, we have successfully implemented our system and reduced the need for manual inspections. The team works in regular sprints and keeps us updated on progress. Their personalized approach, ability to listen, adapt, and continuously refine their methods are truly impressive.”

“Computools’ team truly impressed us with their dedication to the project, their ability to adapt to our processes, and their exceptional hard skills. This allowed us to identify many risks in the initial development stages and address some gaps in our processes. Professionalism, contribution, and flexibility are what define Computools. Based on my experience, I strongly recommend Computools for Dedicated Delivery and outsourcing project services!”

“Computools has delivered a functional solution that helped us increase revenue fivefold, reduce costs, and boost productivity. The team efficiently manages tasks in Jira and keeps us updated through weekly calls. Their productive approach and strong work ethic truly stand out.”

“Computools’ technical knowledge is impressive.They delivered the product on time, within the agreed budget, and fully aligned with our requirements.”

“Thanks to Computools’ efforts, we have seen compliance with deadlines and budget and team scalability as needed. The team has a confident project manager who delivers a professional and organized project. Moreover, Computools has quickly onboarded to the project and delivered fast results.”

“Thanks to the new solution, we’ve significantly reduced manual marketing workflows. Computools manages the project effectively, using Scrum methodology to execute tasks efficiently. Their problem-solving skills and ability to anticipate challenges set them apart from other providers.”

“Computools has successfully delivered everything as planned, adding value to the app. The team is highly approachable, tracks progress, and provides real-time updates via Slack. They maintain smooth communication through email and messaging apps, regardless of time zones.”

“Thanks to Computools, we now have an app that integrates 2,000 users into a single platform, significantly reducing the time spent on data exchange between systems and applications. The team manages our collaboration effectively and quickly adapts to changes. Overall, our experience has been highly successful.”

“Computools has been responsible for creating a novel database and front-end solution, incorporating both the development portal for digital standards and a modern shop for the sale of these standards. Throughout the course of the project, we have been consistently impressed by the professionalism exhibited by the Computools team, as well as their detailed understanding of our client’s processes. Their expertise, commitment to our objectives, and consistent delivery of high-quality work are notable aspects of their service.”

“Thanks to Computools, the client saw a 35% increase in daily active users and a 25% rise in user retention rates. The Android app also saw a 20% reduction in load times. User feedback indicated high user satisfaction; the feedback highlighted the product’s enhanced navigation and content linkage.”

“They are some of the best software developers I ever had the privilege to work with. Among other skills, their project scope and time estimation are very good and when wrong will work around the clock to make the date especially if it has business consequences. Not only are they amazing software developers, but they are also great people to work with. I am in awe seeing their devotion.”

“Computools was selected through an RFP process. They were shortlisted and selected from between 5 other suppliers. Computools has worked thoroughly and timely to solve all security issues and launch as agreed. Their expertise is impressive.”

“After analyzing our requirements, Computools outlined potential solutions and deadlines for each stage. They designed the user flows and defined the user personas. They built the platform infrastructure and oversaw its implementation. Once we finished development, we conducted usability tests to assess their submitted work. Computools led an organized, agile team that adapted to our evolving needs. They listened to our feedback and managed their time well throughout the project.”

“The application perfectly meets the large-scale demands of the project, with the team creating an effective solution that works well and provides the required level of control. They were communicative, responsive, and proactive throughout the project, demonstrating their experience at all times.”
“The Computools team came to us with ideas, and that’s unusual. I’m satisfied that they gave us the right recommendations which are contemporary and relevant for today’s users. Because with other companies on previous projects, it was like pulling teeth to get them to make suggestions. The product received positive feedback even before being implemented and has led to significant customer and revenue growth.”

“We had to meet a significant increase in the development, so we needed to scale up relatively quickly but cost-effectively. The result definitely meets our expectations. The completed project received positive feedback for features and overall design. They’re very organized from a project management perspective and they’re technically competent. We appreciated their innovativeness, professionalism, and great communication skills. ”
“Their team has given us strong learning opportunities, and their developers are accommodating and collaborative.”

“We’re satisfied with the quality of work Computools deliver. They listen and try to understand our needs instead of finding new ways to charge us. We appreciate their transparent work structure. They kept us up-to-date regarding their progress throughout the entire development cycle. Knowing the system’s status throughout the coding process put my mind at ease.”
“They are very accommodating. They have very talented people. I’ve worked with hundreds of overseas developers and it’s not normal to have such excellent overseas developers. I don’t have to babysit Computools. They speak great English. They’ve also really helped with making suggestions on how to improve the product.
When we first launched our product at the beginning of the year, we were at 30,000 users a month and now we’re at 70,000. The bump in users is a result of the increased option rate and the new toys that Computoolls have built for me.”

“Computools developed software for our business to help automate our processes. Their team is very easy to speak to over Skype, where I can speak directly to a designated client manager, project manager, and the development team.”

“They were able to reduce the customer entry acquisition process from 2–3 weeks to 48 hours and have completely optimized all business processes. They’re a trustworthy company, full of integrity and great principles. They also communicate well in spite of the distance and resolve problems quickly.”

“They have a very positive attitude, which I enjoy a lot, and their technical skills are impressive. During this project, I got acquainted with their VP in charge of technical development, and he’s very impressive. Technologically, they are on the cutting edge of what they do. They use a lot of interesting technologies, which is good.”
Our client, a healthcare company, worked with our team to build an opioid monitoring device based on their prototype. The company needed a functional system for patients, and our team helped solve the problem. We leveraged technology to provide scalability, rapid development, data integrity, and seamless integration with external systems. Our team's solution delivered significant benefits to the client by providing a reliable and efficient opioid monitoring device.
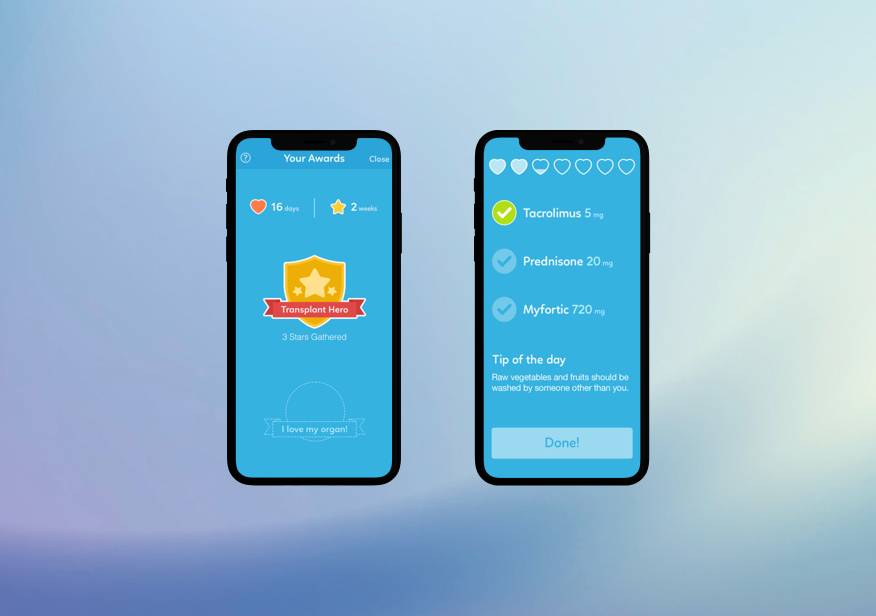
Our client, a healthcare company in the United States, needed an effective tool to help transplant patients adhere to their medication regimens. Our team developed an app with a modernised system that organises medications by time and offers an optimal user experience. The solution resulted in significant improvements for the client, including an increase in average order value, productivity, and revenue.
Since Computools strictly adheres to NDAs with its clients, additional case studies are not publicly available.
Please fill out the contact form, and our experts will respond to your inquiries.
We address key industry pain points such as fragmented data systems, slow documentation cycles, and compliance complexity, by building unified digital platforms that streamline every stage of drug development, production, and distribution.
Yes. Our solutions are built to handle complex, multi-country supply chain operations, offering real-time tracking, automated batch records, and compliance with global serialization and traceability standards.
We create flexible platforms with configurable compliance modules, so when FDA, EMA, or local standards evolve, your system evolves too. No need to rebuild; just update rules and workflows with ease.
Beyond standard functionality, we focus on interoperability, user experience, and future-ready tech, embedding AI, real-time analytics, and scalable cloud infrastructure into every system we deliver.
Absolutely. Whether you’re replacing spreadsheets or upgrading legacy software, we guide you through a structured, low-risk digital transformation, unlocking better insights, automation, and cross-team collaboration.